Category: Pharmaceutical
CSL Behring and CSL Limited
Marburg, Germany
Category: Pharmaceutical
Marburg, Germany
“CSL Behring, a subsidiary of CSL Limited, developed a pandemic vaccine (H1N1) and signed a 180 million dollar contract with the US Department of Health and Human Services (HHS) to provide bulk H1N1 influenza antigen. They use Mitsubishi Electric Iconics Digital Solutions’ software in their FDA-approved filing and packaging facility in Marburg, Germany, to bridge the gap between industrial automation and the medical field.”
CSL Behring, a subsidiary of CSL Limited since 2004, traces its corporate roots to Emil von Behring, the first Nobel Prize recipient for Medicine and Physiology in 1901. As a pioneer in the field of immunology, Behring’s ground-breaking work resulted in the development of vaccinations against diphtheria and tetanus. CSL Biotherapies, also a subsidiary of CSL, operates some of the world’s largest flu vaccine manufacturing facilities for the global market. With more than 80 years of experience in the development and manufacturing of vaccines, and nearly 40 years with flu vaccines, CSL is committed to reliability, safety and quality.
Following the June 2009 World Health Organization announcement of an influenza pandemic, CSL developed a pandemic vaccine (H1N1) and signed a 180 million dollar contract with the US Department of Health and Human Services (HHS) to provide bulk H1N1 influenza antigen. The HHS contract includes the opportunity to use the influenza vaccine dispensing and packaging facilities of CSL Behring in the US and Germany.
“CSL Behring, a subsidiary of CSL Limited, developed a pandemic vaccine (H1N1) and signed a 180 million dollar contract with the US Department of Health and Human Services (HHS) to provide bulk H1N1 influenza antigen. They use Mitsubishi Electric Iconics Digital Solutions’ software in their FDA-approved filing and packaging facility in Marburg, Germany, to bridge the gap between industrial automation and the medical field.”
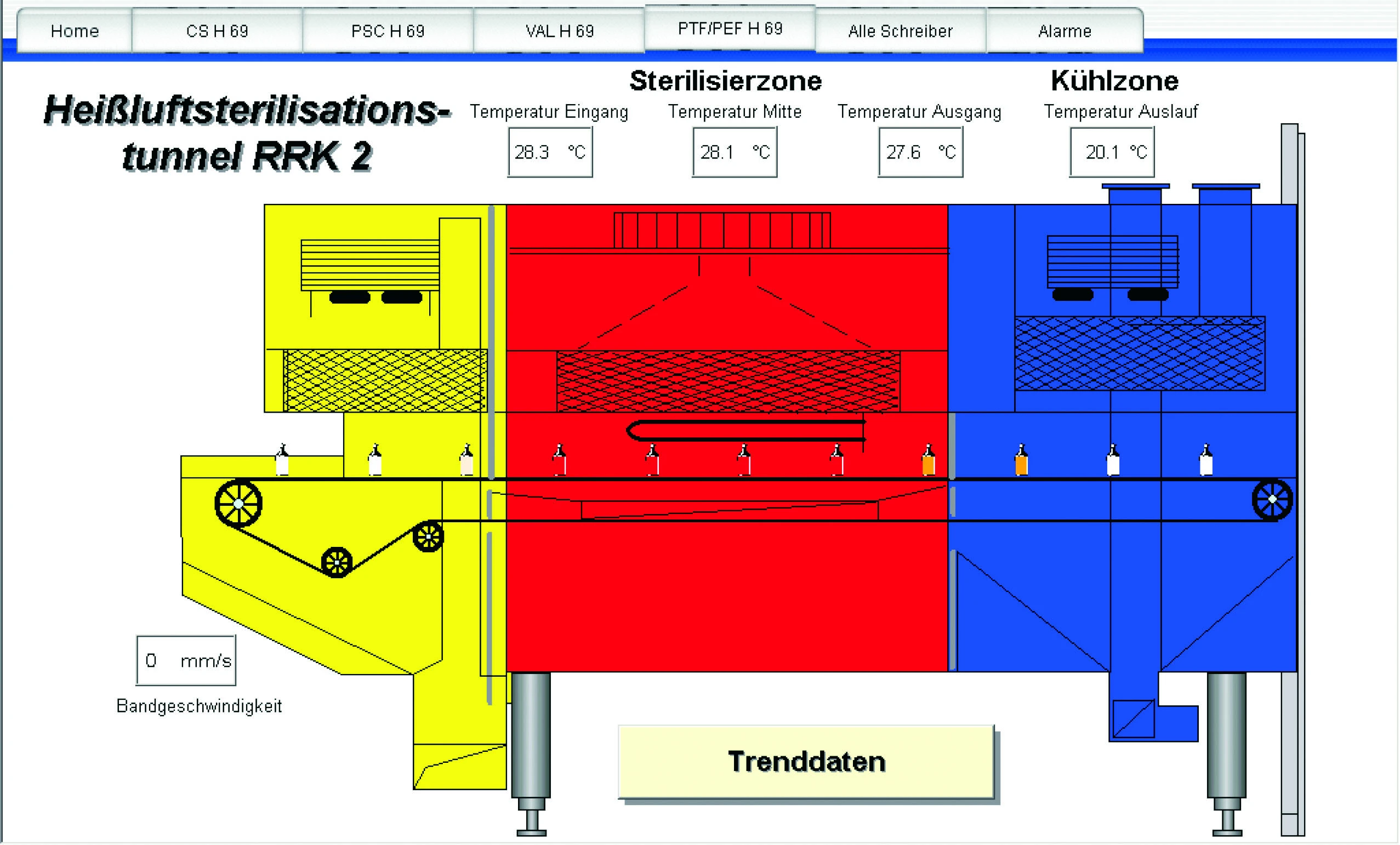 GraphWorX Representation of a Bottling Line
GraphWorX Representation of a Bottling Line
The GENESIS software suite enables rapid and safe production of the H1N1 flu vaccine for the US market in CSL Behring’s FDA-approved filing and packaging facility in Marburg, Germany. DataWorX™, TrendWorX™, WebHMI™, AlarmWorX™ and our OPC DataStore combine to create the solution bridging the gap between industrial automation and the medical field.
 GraphWorX Representation of a Bottling Line
GraphWorX Representation of a Bottling Line
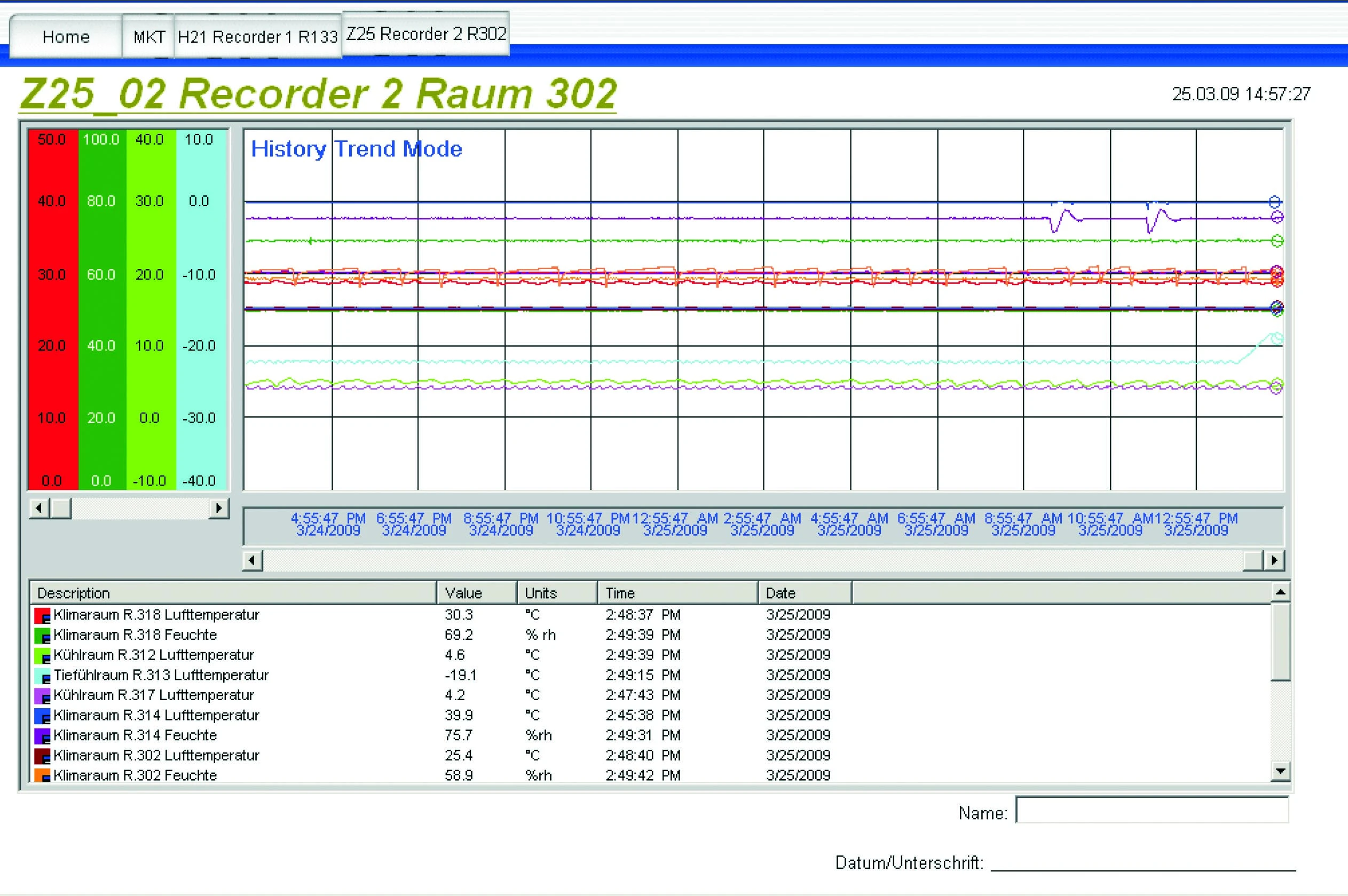 GENESIS Recording Data for CSL Limited
GENESIS Recording Data for CSL Limited
Although their packaging facility is located in Germany, CSL Behring must still meet the recording requirements of the FDA. Specifically, CSL Behring must adhere to the 21 CFR Part 11, which outlines the controls necessary for the regulated industry to utilize electronic records and electronic signatures. CSL Behring requires a system where production data can be logged reliably and stored safely.
Audits, system validations, audit trails, electronic signatures, and documentation of electronic data are subject to inspection for 42 years following production date. GENESIS data and historian applications are designed with these industry-standard regulations in mind.
 GENESIS Recording Data for CSL Limited
GENESIS Recording Data for CSL Limited
 Vials of CSL H1N1 Vaccine
Vials of CSL H1N1 Vaccine
For the past 11 years, Mitsubishi Electric Iconics Digital Solutions (previously ICONICS), in collaboration with System Integrator 8F Computer GmbH, has provided CSL Behring with an automation solution for their bottling facility in Germany. The H1N1 vaccine is dispensed into single use syringes or multi-dose vials in a sterile conveyor line, and then packaged and sent to CSL’s Illinois site for distribution across the US.
TrendWorX facilitates enterprise-wide data collection, logging, charting, reporting and analysis. A DataStore OPC Server stores collected information from proximity sensors, such as bottles in and out, rejects, and total production counts. AlarmWorX improves CSL’s ability to identify, quickly respond to and to document abnormal conditions throughout the bottling process. Internet and Intranet connectivity is provided to GENESIS applications securely through WebHMI.
In total there are 23 servers, running Windows Server, SQL Server, our software and Kepware OPC servers. The total amounts of users who can use the system simultaneously are 109.
 Vials of CSL H1N1 Vaccine
Vials of CSL H1N1 Vaccine
CSL, 8F Computer GmbH and Mitsubishi Electric Iconics Digital Solutions have benefitted from a long lasting relationship, and currently play integral roles in a global environment where the H1N1 influenza vaccine is in high demand. Efficient and safe bottling methods are afforded by our industrial automation solution making for a healthier world.